A Spanish for-profit company collects blood plasma from Canadians, sells products derived from it overseas, and operates four of Canada's eight non-compliant blood facilities. Donors aren't told. The contract is sealed. Two are dead. And the country has done this before.

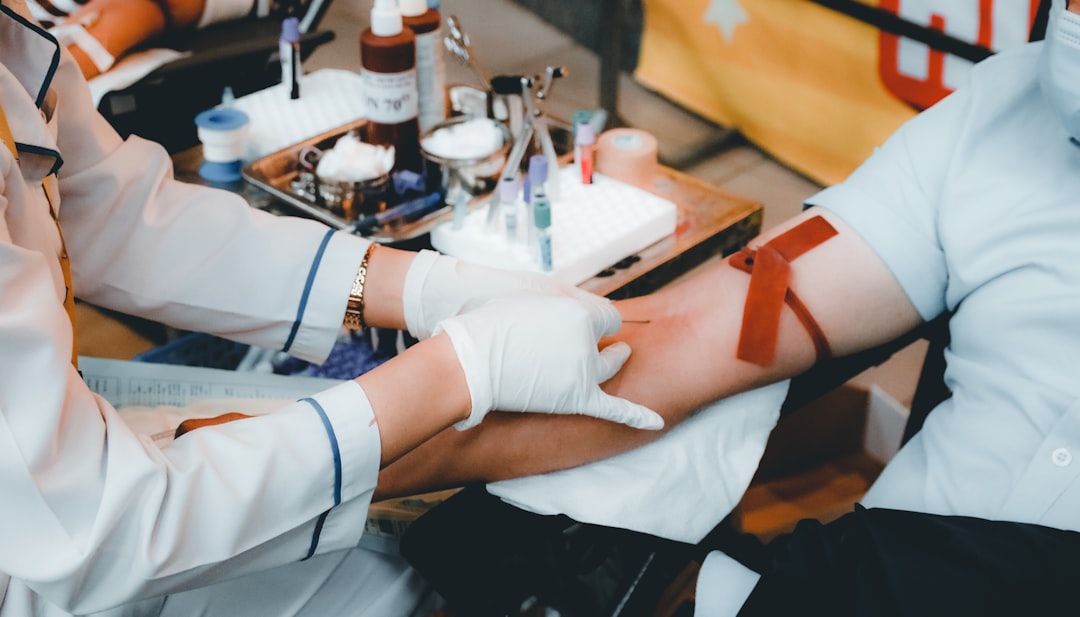
Rodiyat Alabede was 22 years old. She had moved from Nigeria to Winnipeg in 2022 to study at the University of Winnipeg, with the goal of becoming a social worker. Friends remembered her as happy, ambitious, and full of dreams for her new life in Canada. On October 25, 2025, she went to the Grifols Plasma Donation Centre on Taylor Avenue in Winnipeg to donate plasma. She did not come home.1
❝ She was always happy. She had so many dreams, especially when she came to Canada. She held a big place in my heart.
— Mary Ann Chika, friend of Rodiyat Alabede, who died at 22 after donating plasma at a Winnipeg Grifols centreOn January 30, 2026, a second donor died after a fatal adverse reaction at a Winnipeg Grifols centre. Health Canada has confirmed both deaths and says it is investigating, but has not yet linked them publicly to the plasma collection process. The company says it has “no reason to believe” the deaths are connected to its operations. Six months after Alabede’s death, Health Canada has still not released the results of its investigation.2
The company at the centre of the deaths is Grifols — a Spanish multinational pharmaceutical firm that is the largest commercial plasma collector in Canada. Since 2022, Grifols has operated under a 15-year partnership with Canadian Blood Services. The contract has never been made public. Members of Parliament from the House of Commons health committee unanimously voted on March 26 to order CBS and Grifols to produce the agreement by April 10. As of this writing, the contract remains secret.3
Mary Hughes, Grifols’ vice-president of sales in Canada, testified before the health committee on March 26. Under questioning, she made several admissions that should disturb anyone who has ever donated blood in Canada.
First: donors at Grifols centres are not informed that medicines made from their plasma will be sold for profit overseas. They are told the plasma is intended for Canadian Blood Services. They are not told what happens after that.
Second: donors are informed there is a risk of death during the procedure.
Third: when asked whether Grifols was party to any agreement limiting what could be exported from Canadian plasma, Hughes said the terms were “confidential” — even to Parliament.4
As of mid-March 2026, four of the eight blood collection facilities in Canada deemed non-compliant by Health Canada were Grifols centres. The non-compliant Grifols sites are in Calgary, Regina, Saskatoon, and Saint John. A fifth non-compliance report was issued for Grifols’ Canadian head office in Oakville, Ontario. The cited issues included: failing to accurately assess donor suitability, inadequate screening, failing to investigate errors and accidents, failing to determine corrective actions, equipment cleaning failures, and inadequate staff training.5
On April 2, Health Canada placed restrictions on Grifols’ Canadian blood establishment licence, citing “recurring, systemic deficiencies across several sites.” Bloomberg reported the regulator issued non-compliance ratings to the Canadian head office and collection sites in Calgary and Regina.6
To understand why this matters, you have to remember why Canadian Blood Services exists. In the 1980s, Canada experienced what is still considered the worst preventable public health disaster in its history. More than 30,000 Canadians were infected with hepatitis C between 1980 and 1990. Roughly 2,000 were infected with HIV between 1980 and 1985. Approximately 8,000 of those people died or are expected to die as a result. Some of the contaminated blood products were exported, infecting people in Japan, Germany, and Britain.7
In 1993, the federal government established the Royal Commission of Inquiry on the Blood System in Canada — the Krever Commission, headed by Justice Horace Krever. The four-year inquiry produced a report that found misconduct at every level. The Canadian Red Cross, which had operated the blood program for half a century, was stripped of its responsibilities. In 1998, Canadian Blood Services was created to replace it.
The Krever Commission established five principles for Canada’s blood system. The first was that blood and blood products should be collected on a voluntary, unpaid basis. The safety of the blood supply was paramount. These principles aligned with the global consensus established by the European Union, the World Health Organization, and the International Red Cross.
In 2022, Canadian Blood Services signed the 15-year agreement with Grifols to operate paid plasma collection centres across Canada. The decision contradicted the Krever Commission’s founding principles. CBS argued it was unable to meet Canada’s plasma needs through voluntary collection alone — though Héma-Québec, Quebec’s parallel agency, has increased voluntary plasma supply from 15% to 30% in the same period and is on track to reach 50% self-sufficiency without paying donors.8
Donors are not told their plasma is sold overseas. They are told there is a risk of death.
Three provinces — Ontario, British Columbia, and Quebec — have legislation banning paid plasma clinics. Ontario’s Voluntary Blood Donations Act was passed in 2014. The act was enforced until the Ford government allowed Grifols centres to open under an exemption that applies to CBS partnerships. The Ontario Health Coalition has called on the Ford government to enforce its own law and end Grifols’ authorization to collect plasma in the province.9
Manitoba — where both deaths occurred — has no such ban. Health Minister Uzoma Asagwara said this week that “all actions remain on the table” regarding a potential ban. “People who donate plasma need to know their safety will never be put at risk.”
The economic dimension of the contract is staggering. Canadian Blood Services spends approximately $1 billion per year on plasma-derived drugs and related products — about two-thirds of its entire budget. Grifols is the dominant supplier in this market. The company collects plasma from Canadians who are paid for their donations, processes it into medicines, and sells those medicines on the global market. Donors are not informed of the full chain. The contract that governs the relationship is sealed.10
❝ It is disappointing that six months after the first tragic death, Health Canada has still not released the result of its investigation. During the tainted blood scandal it took thousands of deaths and four years of the Krever Commission for governments to act. We can’t wait for years to again protect the safety of our blood system.
— Geoff Cain, retired CBS staffer and chair of the Ontario Health Coalition’s Committee to Protect Ontario’s Blood SystemConservative MP Dan Mazier raised the issue in committee, asking why Canadians should donate plasma to a system that may be selling derivatives overseas without their knowledge. His questions were echoed by NDP MP and others. The committee’s unanimous order for the contract to be produced by April 10 was the rare bipartisan moment in Parliament — a sign that even members of the governing party recognize the secrecy is unsustainable.
The Canadian Health Coalition has called for the resignation of the CBS CEO and board of directors. The Centre for Policy Alternatives has called for a full judicial inquiry under Manitoba’s Fatality Inquiries Act. Health policy experts have noted that during the original tainted blood scandal, it took thousands of deaths and four years of the Krever Commission for governments to act. “We can’t wait for years to again protect the safety of our blood system,” said Geoff Cain, a retired CBS staffer and chair of the Ontario Health Coalition’s Committee to Protect Ontario’s Blood System.11
Two people are dead. Four of Canada’s eight non-compliant blood collection facilities belong to a Spanish for-profit corporation. Donors are not told that medicines made from their plasma will be sold overseas. They are told there is a risk of death. The contract between Canadian Blood Services and Grifols has been secret for four years and remains secret today, despite a unanimous parliamentary order to produce it. Three provinces have laws banning paid plasma collection. One of them — Ontario — is being circumvented by a regulatory exemption. The agency that operates the contract was created in 1998 specifically to prevent the kind of for-profit, opaque, internationally-traded blood system that produced Canada’s worst public health disaster. Twenty-eight years later, that system is back. The first two deaths have already happened. The country has done this before. The question is whether anyone in power remembers what came after.
Every source. Every contradiction. Yours to share.